NIRS - Near InfraRed Spectroscopy
The innovative method for performance diagnostics and training control
How does NIRS work?
NIRS sends light near the infrared spectrum into the tissue and measures the reflection of the light. The Moxy has a "transmitter" that sends the light about 15mm deep through the skin as well as the subcutaneous fat tissue into the muscles and 2 "receivers" that measure the reflections.
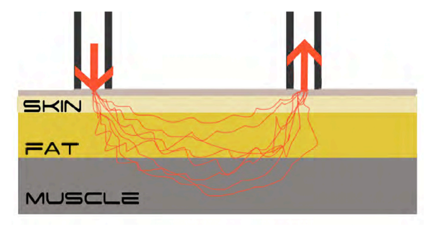
The amount of reflected light depends mainly on the light-absorbing molecules in the examined tissue. In the case of skeletal muscle, these are essentially the iron compounds of haemoglobin and myoglobin, which reflect more or less light depending on their connection with oxygen. Accordingly, NIRS can be used to measure the concentration and oxygen saturation of haemoglobin and myoglobin in the tissue under investigation, with the haemoglobin located in the microvessels around the muscle and the myoglobin in the muscle itself. Factors influencing the measurement are haemoglobin concentration of the blood, capillarisation, muscle fibre composition, subcutaneous fat thickness and skin pigmentation. The higher the haemoglobin concentration of the blood and the capillarisation, the more haemoglobin in the blood and correspondingly more oxygen can be transported to the muscle. Slow muscle fibres have a higher concentration of myoglobin than fast muscle fibres. Thus, the measured haemoglobin is higher with a high proportion of slow fibres than with a high proportion of fast fibres. The thickness of the subcutaneous fat as well as the thickness of the skin pigmentation influence how much light can reach the muscle and its surrounding vessels. The thicker the subcutaneous fatty tissue, the fewer blood vessels and muscles are reached by the emitted light. The stronger the skin pigmentation, the less light penetrates the skin.
What does NIRS measure?
Strictly speaking, NIRS measures the concentration and oxygen saturation of haemoglobin and myoglobin.
There are various statements on the ratio of the share of the NIRS signal between haemoglobin (Hb) and myoglobin (Mb). Some studies assume a predominant share of Hb (Mancini 1997b). Others assume a predominant share of Mb (Marcinek et al. 2007). Regardless of the share in the NIRS signal, the shares of Hb and Mb show intraindividual variations. For example, the concentration of Mb is higher in type I muscle fibres than in type II muscle fibres (van Beek-Harmsen et al. 2004; Nemeth and Lowry 1984), which means that the proportion of the total signal is influenced by the distribution of the muscle fibres. Furthermore, it has been shown that the influence of Hb on the NIRS signal is a function of microvascular volume as well as microvascular haematocrit (Barstow 2019). Thus, the better the capillarisation, the greater the contribution of Hb to the NIRS signal. This correlation is also or especially reflected under stress. Due to the increasing blood flow under stress, the proportion of Hb in the NIRS signal increases (Davis and Barstow 2013). Thus, the proportions of Hb and Mb in the NIRS signal differ not only statically (muscle fibre composition, capillarisation), but also dynamically (activity).
Due to common nomenclature and to simplify the presentation, we use the reference to Hb in the following, being aware that it is always the sum of Hb + Mb.
Four basic variables can be derived from the measurement: Oxygen saturation (SmO2), oxygenated Hb (oxy-Hb), non-oxygenated Hb (deoxy-Hb), total Hb (tHb), and variables derived from these. The basic variables are briefly explained below.
- Oxygen saturation (SmO2)
Oxygen saturation represents the concentration of oxygenated haemoglobin in the tissue under examination. It thus represents the balance between oxygen supply and oxygen consumption of the skeletal muscle. - Total haemoglobin (tHb)
Changes in total haemoglobin represent changes in capillary haematocrit and thus serve as an indicator of local blood flow (Barstow 2019). Thus, an increase in tHb represents increased blood flow and vice versa. A decrease in tHb can be triggered, for example, by a low cadence in combination with high torque and the resulting interruption of local blood flow. - Oxyhaemoglobin (O2Hb)
Oxyhaemoglobin describes the oxygen-enriched haemoglobin and represents the local oxygen supply of the skeletal muscle. It is therefore composed of the current oxygen saturation and the change in total haemoglobin. - Deoxyhaemoglobin (HHb)
Deoxyhaemoglobin is the haemoglobin that is not enriched with oxygen. It reflects the metabolic oxygen demand of the muscle (Wang et al. 2006).
Numerous studies have demonstrated that NIRS technology can be used to determine individual anaerobic threshold, lactate threshold, respiratory compensation point (RCP) and lactate balance (Bellotti et al. 2013; Fontana et al. 2015; Keir et al. 2015; Wang et al. 2006).
In addition, several studies have demonstrated that there is a close correlation between NIRS parameters and the high-energy phosphates (e.g. creatine phosphate) (McCully et al. 1994; Ryan et al. 2013).
Thus, NIRS technology can be seen as a reliable measurement method that allows numerous conclusions to be drawn about the underlying physiological processes.
Literature
Bellotti, Cecilia; Calabria, Elisa; Capelli, Carlo; Pogliaghi, Silvia (2013): Determination of maximal lactate steady state in healthy adults: can NIRS help? In Medicine and science in sports and exercise 45 (6), pp. 1208-1216. DOI: 10.1249/MSS.0b013e3182828ab2.
Davis, Michelle L.; Barstow, Thomas J. (2013): Estimated contribution of hemoglobin and myoglobin to near infrared spectroscopy. In Respiratory physiology & neurobiology 186 (2), pp. 180-187.
Fontana, Federico Y.; Keir, Daniel A.; Bellotti, Cecilia; Roia, Gabriela F. de; Murias, Juan M.; Pogliaghi, Silvia (2015): Determination of respiratory point compensation in healthy adults: Can non-invasive near-infrared spectroscopy help? In Journal of science and medicine in sport 18 (5), pp. 590-595. DOI: 10.1016/j.jsams.2014.07.016.
Keir, Daniel A.; Fontana, Federico Y.; Robertson, Taylor C.; Murias, Juan M.; Paterson, Donald H.; Kowalchuk, John M.; Pogliaghi, Silvia (2015): Exercise Intensity Thresholds: Identifying the Boundaries of Sustainable Performance. In Medicine and science in sports and exercise 47 (9), pp. 1932-1940. DOI: 10.1249/MSS.00000000000613.
Mancini, D. (1997a): Application of near infrared spectroscopy to the evaluation of exercise performance and limitations in patients with heart failure. In Journal of biomedical optics 2 (1), pp. 22-30. DOI: 10.1117/12.263747.
Mancini, D. (1997b): Application of near infrared spectroscopy to the evaluation of exercise performance and limitations in patients with heart failure. In Journal of biomedical optics 2 (1), pp. 22-30. DOI: 10.1117/12.263747.
Marcinek, David J.; Amara, Catherine E.; Matz, Kimberly; Conley, Kevin E.; Schenkman, Kenneth A. (2007): Wavelength shift analysis: a simple method to determine the contribution of hemoglobin and myoglobin to in vivo optical spectra. In Applied spectroscopy 61 (6), pp. 665-669.
McCully, K. K.; Iotti, S.; Kendrick, K.; Wang, Z.; Posner, J. D.; Leigh, J.; Chance, B.. (1994): Simultaneous in vivo measurements of HbO2 saturation and PCr kinetics after exercise in normal humans. In Journal of applied physiology (Bethesda, Md. : 1985) 77 (1), pp. 5-10. DOI: 10.1152/jappl.1994.77.1.5.
Nemeth, P. M.; Lowry, O. H. (1984): Myoglobin levels in individual human skeletal muscle fibers of different types. In The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society 32 (11), pp. 1211-1216. DOI: 10.1177/32.11.6491255.
Ryan, Terence E.; Southern, W. Michael; Reynolds, Mary Ann; McCully, Kevin K. (2013): A cross-validation of near-infrared spectroscopy measurements of skeletal muscle oxidative capacity with phosphorus magnetic resonance spectroscopy. In Journal of applied physiology (Bethesda, Md. : 1985) 115 (12), pp. 1757-1766. DOI: 10.1152/japplphysiol.00835.2013.
van Beek-Harmsen, Brechje J.; Bekedam, Martijn A.; Feenstra, H. Maria; Visser, Frans C.; van der Laarse, Willem J. (2004): Determination of myoglobin concentration and oxidative capacity in cryostat sections of human and rat skeletal muscle fibres and rat cardiomyocytes. In Histochemistry and cell biology 121 (4), pp. 335-342.
Wang, Lixin; Yoshikawa, Takahiro; Hara, Taketaka; Nakao, Hayato; Suzuki, Takashi; Fujimoto, Shigeo (2006): Which common NIRS variable reflects muscle estimated lactate threshold most closely? In Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabol ism 31 (5), pp. 612-620. DOI: 10.1139/h06-069.
